Home
TVAX Biomedical is developing a paradigm-shifting cancer treatment that offers the promise of improved clinical outcomes and low toxicity and the potential for fundamentally changing the way cancer is treated.
TVAX Immunotherapy® is a unique personalized “killer” T cell treatment. TVAX Immunotherapy employs the natural ability of T cells to kill cancer cells. Activated “killer” T cells have demonstrated the ability to effectively treat numerous cancers without many of the undesirable side effects associated with radiation and chemotherapy. TVAX is distinct from other cancer immunotherapy companies in its use of ex vivo-activated cancer antigen-specific “killer” T cells as the active agent in its treatment. To learn more, click here.
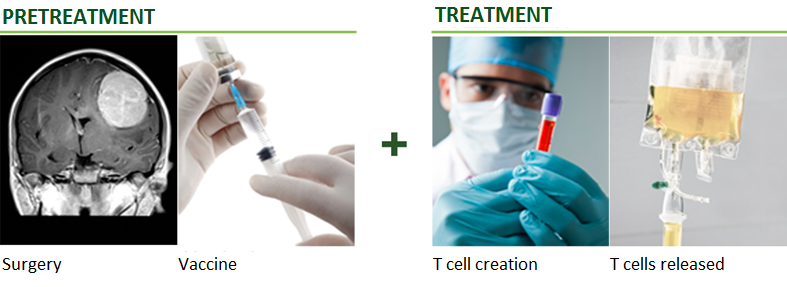
TVAX’s lead candidate (TVI-Brain-1) is currently being evaluated for the treatment of brain cancer, specifically glioblastoma. The company has been granted Fast Track Designation by the United States Food and Drug Administration (FDA) to conduct a pivotal Phase 2b trial for TVI-Brain-1 to support the therapy’s potential FDA approval. Additionally, the company received a $2 million grant from the FDA office of orphan products. To learn more, click here.

